Why does it still show air pollution when the sky is clear in summer?
my country's Air Quality Index (AQI) is composed of six indicators: fine particulate matter (PM2.5), particulate matter (PM10), ozone (O3), sulfur dioxide (SO2), nitrogen dioxide (NO2), and carbon monoxide (CO).
After years of hard work, except for ozone, the other five indicators have declined year by year. Summer is the season of high incidence of ozone pollution, and the clear sky means that PM10 and PM2.5 are low. If air pollution is still displayed at this time, it is most likely because the O3 in the air exceeds the standard.
What is ozone (O3)?
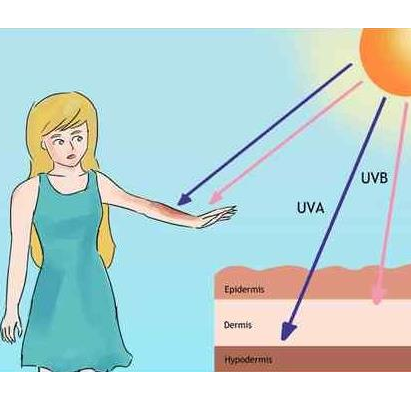
O3 is a trace gas in the earth's atmosphere. It is formed when oxygen molecules in the atmosphere are decomposed into oxygen atoms by solar radiation, and then the oxygen atoms combine with the surrounding oxygen molecules. It contains 3 oxygen atoms, and the chemical formula is O3. More than 90% of O3 in the atmosphere exists in the stratosphere, 10 to 50 km from the ground. It can effectively block the ultraviolet radiation of sunlight and protect human beings and the environment.
However, if the concentration of O3 near the ground is relatively high, it will have harmful effects on human health and the ecological environment. For example, a higher concentration of O3 has an irritating effect on the eyes and respiratory tract, and also affects lung function. A higher concentration of O3 is also harmful to plants. harmful.
How is O3 formed near the ground?
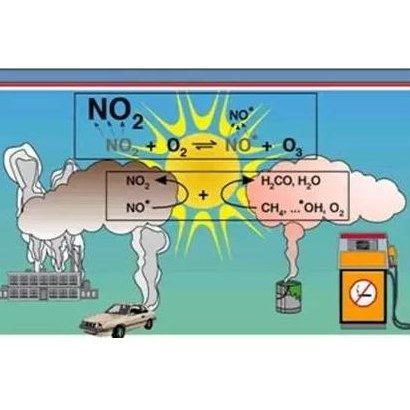
O3 in the near-surface (troposphere) atmosphere is mainly produced by photochemical reaction processes, that is, O3 is a "secondary pollutant". Precursors such as nitrogen oxides (NOx) and volatile organic compounds (VOCs) emitted by industrial enterprises undergo a series of complex photochemical reactions under the irradiation of sunlight (ultraviolet rays) to produce O3 pollutants.
The sources of NOx are wide, but they are basically man-made emissions, mainly from motor vehicle exhaust, fossil fuel combustion, and industrial production processes; VOCs come from more extensive sources, including petrochemical, pharmaceutical, chemical, furniture, auto repair, printing and other industries and industries. Exhaust emissions from enterprises also include volatilization of oil and gas from motor vehicles and gas stations, as well as volatilization of organic matter such as cooking fumes, dry cleaners, and hairdressers. Therefore, O3 pollution also presents the characteristics of wide coverage and large influence.
Is O3 harmful to the human body?
Low concentrations of O3 can be sterilized, but excessive O3 is an invisible killer. Its hazards include:
(1) Stimulate the human respiratory tract, causing sore throat, chest tightness and cough, causing bronchitis and emphysema;
(2) cause neurotoxicity, dizziness, headache, vision loss, memory loss;
(3) It has a destructive effect on vitamin E in human skin, causing human skin to wrinkle and dark spots;
(4) It will also destroy the immune function of the human body, induce chromosomal lesions of lymphocytes, accelerate aging, and cause pregnant women to have deformed children.
What is the hazardous concentration of O3?
To speak of toxicity aside from metrology is pseudoscience.
When the concentration of O3 is 20 ~ 40 μg/L, there is no adverse reaction in the human body; when the concentration is 100 ~ 140 μg/L, there is a feeling of irritation to the mucous membranes of the eyes, nose and throat; when the concentration is 140 ~ 160 μg/L, dry mouth occurs Dry tongue, sore throat, cough and other symptoms; when the concentration of O3 exceeds 10,000 μg/L, it will cause great harm to the human body.
According to my country's "Ambient Air Quality Standard" (GB3095-2012), the concentration limit of O38 hours should be ≤ 160μg/L in residential areas, mixed areas of commercial traffic and residents, cultural areas, industrial areas and rural areas, and the concentration limit of O31 hours should be ≤ 200μg/L. When this concentration is exceeded, the environmental department will act quickly to find and reduce pollution sources and protect people's health.
Because the odor of O3 is very strong, people can feel it when the concentration exceeds 200 μg/L. Therefore, O3 has been used in the world for more than 100 years, and there has been no report of death due to O3 poisoning.
What time of year does O3 have the highest concentration?
From the perspective of the whole year, the occurrence of O3 pollution generally starts from April every year and lasts until October, of which the concentration is relatively high from June to August. From the perspective of one day, as the temperature rises in the morning, the ultraviolet radiation increases, and the O3 concentration continues to increase, and the high value continues from 1 to 3 p.m. in the afternoon, and the O3 concentration gradually decreases as the radiation weakens around 5 p.m. in the evening. Therefore, in general, the O3 concentration is highest around 3:00 pm in summer.
What is the reason for the high O3 concentration in summer?
O3 pollution has special conditions for its generation. Under the conditions of high temperature, sufficient sunshine and dry air, VOCs and NOx in the air "meet" to produce photochemical reactions, which are easy to generate O3 pollution.
The formation of O3 in summer can be described as "the right time and place": strong sunshine, high temperature, less cloud cover, weak wind, and sufficient "raw materials".
How can individuals protect against O3?
Different from PM2.5, O3 has smaller molecules and cannot be intercepted by masks, gas masks, etc.
Therefore, individuals should do "one check and two reductions" during the high temperature season in summer. A check, refers to the high temperature days in summer, check the O3 concentration broadcast on that day. Second, when the O3 concentration exceeds the standard, the outdoor exercise time should be reduced to prevent the human body from directly contacting high concentrations of O3; the number of indoor and outdoor ventilation should be reduced to prevent O3 pollution from entering the room.
Will O3 wander around?
O3 pollution can be prevented and controlled, but it is also difficult to prevent and control. According to scientific research and expert judgment, O3 pollution has certain regional transmission characteristics, it will stray everywhere, and it will also flow everywhere. The emission of VOCs and NOx in a single city does not necessarily appear in a specific emission when O3 pollution is generated. region and city.
Therefore, in the event of high temperature and dry weather, it is necessary to implement social linkage and regional joint control to fight the tough battle of O3 pollution control.