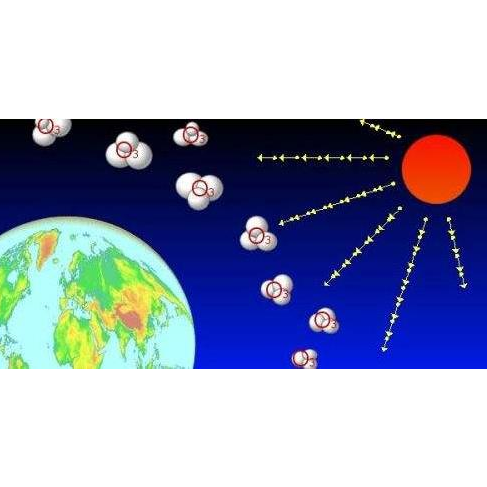
Ozone is a naturally occurring component in fresh air. It can be produced by the reaction of sunlight's ultraviolet rays with the earth's atmosphere, which protects the earth's ozone layer, or it can be artificially created by an ozone machine. The ozone molecule has three oxygen atoms, and the oxygen molecule has only two. Ozone is a reactive and unstable gas with a very short half-life before being converted back to oxygen. Ozone is the most powerful and fastest-acting oxidant that humans can make, oxidizing all bacteria, molds, yeast spores, organic matter, and viruses.
For oxygen to form ozone, some energy must be used. In this process, either electrical releasers such as cadmium-type ozone generators (corona discharge - mimics lightning) or ultraviolet radiation such as UV ozone generators (ultraviolet rays that mimic sunlight) are used. In addition to these commercial methods, ozone can also be obtained through electrolysis as well as chemical reactions.